A Resource of High-quality and Versatile Nanobodies for Drug Delivery
Shen Z, Xiang Y, Vergara S, Chen A, Xiao Z, Santiago U, Jin C, Sang Z, Luo J, Chen K, Schneidman-Duhovny D, Camacho C, Calero G, Hu B, Shi Y
iScience
2021 Aug 21
Chitinase-3-like 1 Protein Complexes Modulate Macrophage-mediated Immune Suppression in Glioblastoma
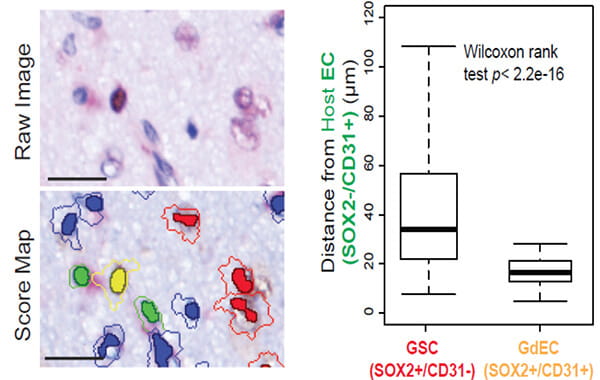
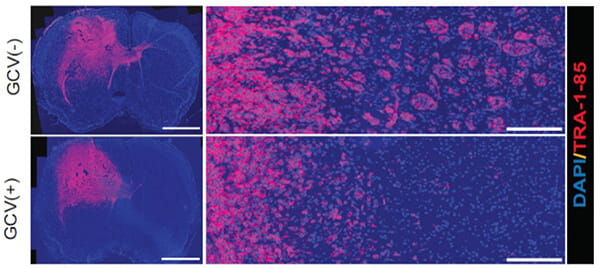
Chen A, Jiang Y, Li Z, Wu L, Santiago U, Zou H, Cai C, Sharma V, Guan Y, McCarl LH, Ma J, Wu YL, Michel J, Shi Y, Konnikova L, Amankulor NM, Zinn PO, Kohanbash G, Agnihotri S, Lu S, Lu X, Sun D, Gittes GK, Wang Q, Xiao X, Yimlamai D, Pollack IF, Camacho CJ, Hu B
The Journal of Clinical Investigation
2021 Aug 16
ZFTA–RELA Dictates Oncogenic Transcriptional Programs to Drive Aggressive Supratentorial Ependymoma
Arabzade A, Zhao Y, Varadharajan S, Chen HC, Jessa S, Rivas B, Stuckert AJ, Solis M, Kardian A, Tlais D, Golbourn BJ, Stanton AJ, Chan YS, Olson C, Karlin KL, Kong K, Kupp R, Hu B, Injac SG, Ngo M, Wang PR, De León LA, Sahm F, Kawauchi D, Pfister SM, Lin CY, Hodges HC, Singh I, Westbrook TF, Chintagumpala MM, Blaney SM, Parsons DW, Pajtler KW, Agnihotri S, Gilbertson RJ, Yi J, Jabado N, Kleinman CL, Bertrand KC, Deneen B, Mack SC
Cancer Discovery
2021 Sep 11
Therapy-Induced Transdifferentiation Promotes Glioma Growth Independent of EGFR Signaling
Oh H, Hwang I, Jang JY, Wu L, Cao D, Yao J, Ying H, Li JY, Yao Y, Hu B, Wang Q, Zheng H, Paik J
Cancer Research
2021 Mar 15
Telomere Dysfunction Activates YAP1 to Drive Tissue Inflammation
Chakravarti D, Hu B, Mao X, Rashid A, Li J, Li J, Liao WT, Whitley EM, Dey P, Hou P, LaBella KA, Chang A, Wang G, Spring DJ, Deng P, Zhao D, Liang X, Lan Z, Lin Y, Sarkar S, Terranova C, Deribe YL, Blutt SE, Okhuysen P, Zhang J, Vilar E, Nielsen OH, Dupont A, Younes M, Patel KR, Shroyer NF, Rai K, Estes MK, Wang YA, Bertuch AA, DePinho RA
Nature Communications
2020 Sep 21
Tumor Evolution of Glioma-Intrinsic Gene Expression Subtypes Associates with Immunological Changes in the Microenvironment
Wang Q, Hu B, Hu X, Kim H, Squatrito M, Scarpace L, deCarvalho AC, Lyu S, Li P, Li Y, Barthel F, Cho HJ, Lin YH, Satani N, Martinez-Ledesma E, Zheng S, Chang E, Sauvé CG, Olar A, Lan ZD, Finocchiaro G, Phillips JJ, Berger MS, Gabrusiewicz KR, Wang G, Eskilsson E, Hu J, Mikkelsen T, DePinho RA, Muller F, Heimberger AB, Sulman EP, Nam DH, Verhaak RGW
Cancer Cell
2017 Jul 10
PAF Promotes Stemness and Radioresistance of Glioma Stem Cells
Ong DST, Hu B, Ho YW, Sauve, GC, Bristow CA, Wang Q, Multani SA, Chen P, Nezi L, Jiang S, Gorman CE, Monasterio, MM, Koul D, Marchesini M, Colla S, Jin EJ, Sulman EP, Lang FF, Spring DJ, Yung AW, Verhaak RGW, Chin L, Wang YA, and DePinho RA
Proceedings of the National Academy of Sciences of the USA
2017 Oct 24
QKI Deficiency Maintains Stemness of Glioma Stem Cells in Suboptimal Environment by Downregulating Endolysosomal Degradation
Shingu T, Ho A, Yuan L, Zhou X, Dai C, Zheng S, Wang Q, Zhong Y, Chang, Q, Horner J, Liebelt BD, Yao Y, Hu B, Chen Y, Fuller GN, Verhaak RGW, Heimberger AM, Hu J
Nature Genetics
2017 Jan
Epigenetic Activation of WNT5A Drives Glioblastoma Stem Cell Differentiation and Invasive Growth
Hu B, Wang Q, Wang YA, Hua S, Sauve, GC, Ong DS, Zheng, DL, Chang Q, Ho YW, Monasterio, MM, Lu X, Zhong Y, Zhang J, Deng P, Tan Z, Wang G, Liao, W, Corley LJ, Yan H, Zhang J, You, Y, Liu N, Cai L, Finocchiaro G, Phillips JJ, Berger MS, Spring DJ, Hu J, Sulman EP, Fuller GN, Chin L, Verhaak RGW, DePinho RA
Cell
2016 Nov 17
Targeting YAP-Dependent MDSC Infiltration Impairs Tumor Progression
Wang G, Lu X, Dey P, Deng P, Wu CC, Jiang S, Fang Z, Zhao K, Konaparthi R, Hua S, Zhang J, Li-Ning-Tapia EM, Kapoor A, Wu CJ, Patel NB, Guo Z, Ramamoorthy V, Tieu TN, Heffernan T, Zhao D, Shang X, Khadka S, Hou P, Hu B, Jin EJ, Yao W, Pan X, Ding Z, Shi Y, Li L, Chang Q, Troncoso P, Logothetis CJ, McArthur MJ, Chin L, Wang YA, DePinho RA
Cancer Discovery
2016 Jan
From the Cover: Neutralization of Terminal Differentiation in Gliomagenesis
Hu J, Ho AL, Yuan L, Hu B, Hua S, Hwang SS, Zhang J, Hu T, Zheng H, Gan B, Wu G, Wang YA, Chin L, DePinho RA
Proceedings of the National Academy of Sciences of the USA
2013 Sep 3
Passenger Deletions Generate Therapeutic Vulnerabilities in Cancer
Muller FL, Colla S, Aquilanti E, Manzo VE, Genovese G, Lee J, Eisenson D, Narurkar R, Deng P, Nezi L, Lee MA, Hu B, Hu J, Sahin E, Ong D, Fletcher-Sananikone E, Ho D, Kwong L, Brennan C, Wang YA, Chin L, DePinho RA
Nature
2012 Aug 15
STAR RNA-binding Protein Quaking Suppresses Cancer via Stabilization of Specific miRNA
Chen AJ, Paik JH, Zhang H, Shukla SA, Mortensen R, Hu J, Ying H, Hu B, Hurt J, Farny N, Dong C, Xiao Y, Wang YA, Silver PA, Chin L, Vasudevan S, Depinho RA
Genes & Development
2012 Jul 1